What are silicon carbide and alumina?
Silicon Carbide

Silicon carbide or SiC is a chemical produced from the one-to-one binding of carbon (C) and silicon (Si), which is seldom found in nature.
- Applications
- Silicon carbide (SiC) has high hardness, outstanding heat resistance and durability. Therefore, it is used for grinding stones, coated abrasives, refractory products and heating elements. In addition, SiC is more resistant to chemical corrosion, such as oxidation, than other non-oxide ceramics; consequently, it is used for crucibles, parts for kiln burning, mechanical seals, and parts materials to produce semiconductors. It also used in products such as heat sheets for thermolysis due to its high thermal conductivity.
- Features
-
■Hardness
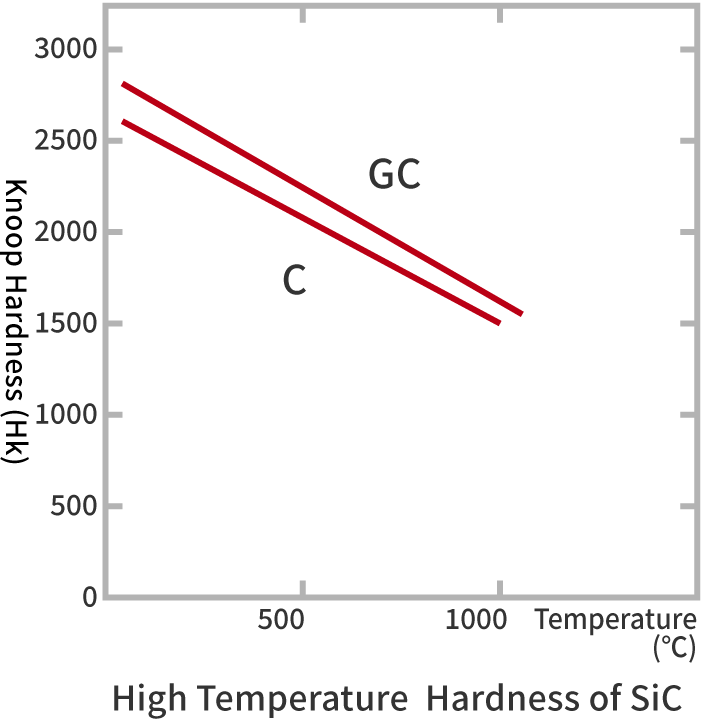
Silicon carbide has a crystal structure that is very similar to that of diamond, and is a very hard chemical compound with the feature of being difficult to deform by pressing.
■Heat Resistance
Silicon carbide has a high decomposition temperature and outstanding heat resistance.
■Heat Conductibility
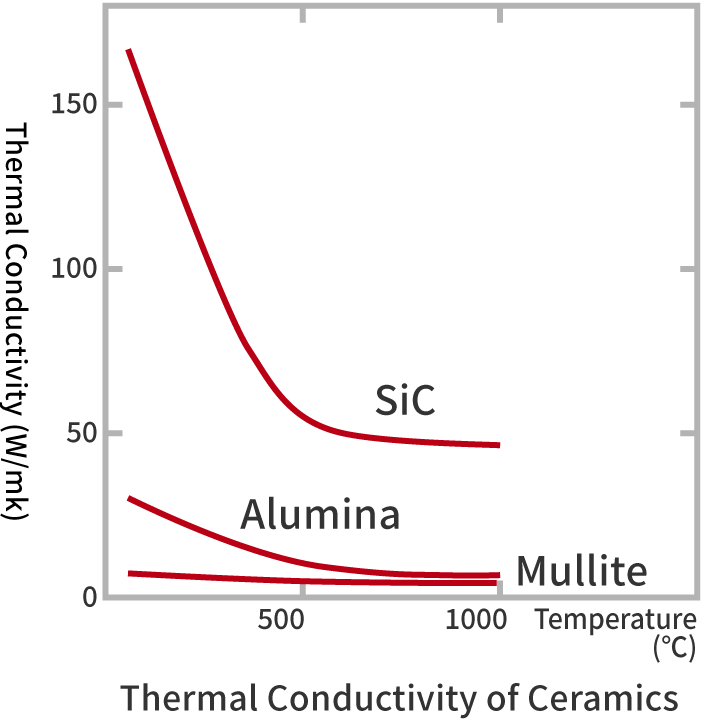
Silicon carbide is a good conductor of heat.
■Chemical Resistance
Silicon carbide is chemically stable and has high corrosion resistance, which makes it hard to corrode from exposure to alkali or acid.
<“Ceramic New Materials of SiC System” compiled by the No. 124 Committee, High Temperature Ceramic Materials, Japan Society for the Promotion of Science>
Alumina

Alumina(Al2O3) is a chemical compound of aluminum and oxygen, generally called aluminum oxide. Aluminum is the third most abundant element on Earth after oxygen and silicon.
- Applications
- Because high-density sintered body with high purity and high dimensional accuracy can be relatively easily manufactured with alumina (Al2O3), it is one of the most versatile materials in application among the ceramics. It is not only used for mechanical applications with mechanical strength, abrasion resistance, and corrosion resistance, but is also widely used for electronic engineering applications such as GIS as well as insulator components and integrated circuit boards using electric insulation, and optical applications that make use of translucency based on a special production method.
- Features
-
■Hardness
Alumina is the hardest of the oxides.
■Heat Conductibility
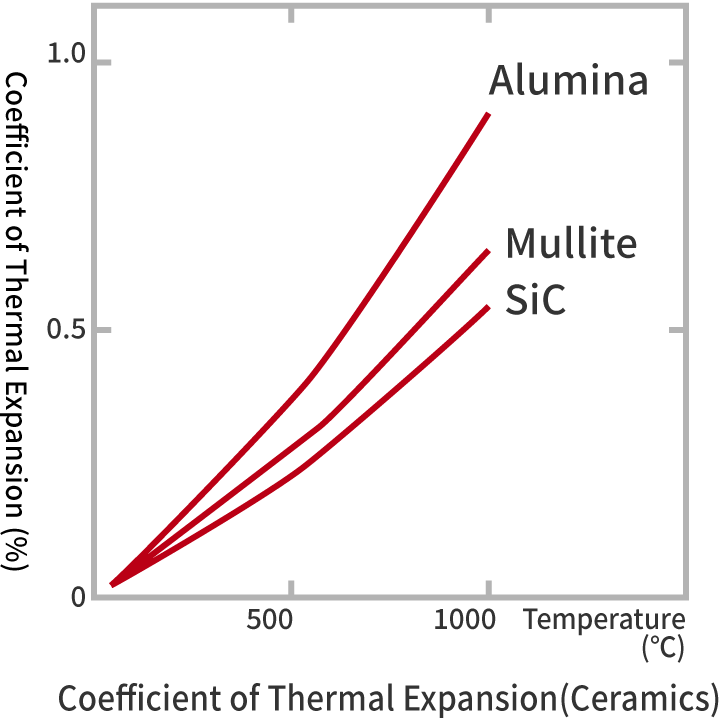
Alumina is a substance whose heat conductivility is relatively high. On the other hand, its coefficient of thermal expansion is large, and the impact resistance is low compared to silicon nitride or silicon carbide.
■High Electric Insulating Characteristic
Alumina sintered body with less soda has an electrical resistance of over 1015 Ωcm.
■Corrosion Resistance
Alumina is chemically stable and shows excellent corrosion resistance against chemicals such as acids.
<“Engineering Ceramics” page 22, by Hiroshi Abe, Minoru Kawai, Takashi Kanno, Keiichiro Suzuki (Gihodo Shuppan Co. Ltd., 1st ed., 1984)>
Features
Features
| Classification | Kind | JIS Mark | Property | Use | Nisso Rundum |
|---|---|---|---|---|---|
| Silicon Carbide Grinding Material | Black Silicon Carbide Grinding Material | C | High hardness and sharp cutting edge | Precision grinding of nonmetals, non-ferrous metals, and cast iron | NC, RC |
| Green Silicon Carbide Grinding Material | GC | Higher hardness and friability than C | Sintered hard alloy grinding | NG, GMF | |
| Alumina Grinding Material | Brown Alumina Grinding Material | A | Al2O3 with solid-soluted TiO2, it can absorb impacts and is strong | Free grinding of general steel Precision grinding of raw steel |
ー |
| White Alumina Grinding Material | WA | It does not contain TiO2, has high friability and sharp cutting edge | Precision and light grinding of alloy steel and tool steel | 50A, 50R, LA, BL | |
| Rose-Pink Alumina Grinding Material | PA | Al2O3 with solid-soluted Cr2O3, allows it to have friability as well as toughness | Precision grinding of alloy steel and special steel | 50P | |
| Mono-Crystalline Alumina Grinding Material | HA | Strong nature and excellent grindability | Grinding of multipurpose screws, gears, and tools of alloy steel | ー |
Physical Properties
| Item | Silicon Carbide | Alumina | |
|---|---|---|---|
| True Density (g/cm3) | 3.21 | 3.98 | |
| Knoop Hardness (Hk) | ~2,600~ | ~2,000~ | |
| Melting Point (℃) | ー (Be sublimed at a temperature of 2545 degree) |
2,050 | |
| Specific Heat (kJ/kg・K) | 0.63~1.26 | 0.92~1.26 | |
| Heat Conductivity (W/mK) | RT | 167.6 | 30.2 |
| 400℃ | 67.0 | 13.0 | |
| 1000℃ | 46.9 | 6.3 |
|
| Coefficient of Thermal Expansion (×10-6/K) | RT | 3.8 | 6.9 |
| 400℃ | 4.1 | 7.8 | |
| 1000℃ | 4.8 | 8.3 | |
Thermal Properties, etc.